Gray-market peptides near a regulatory inflection.
Policy shift. The FDA is reconsidering its 2023 ban on certain injectable peptides — with advisory review starting in July, opening the door to compounding if cleared.
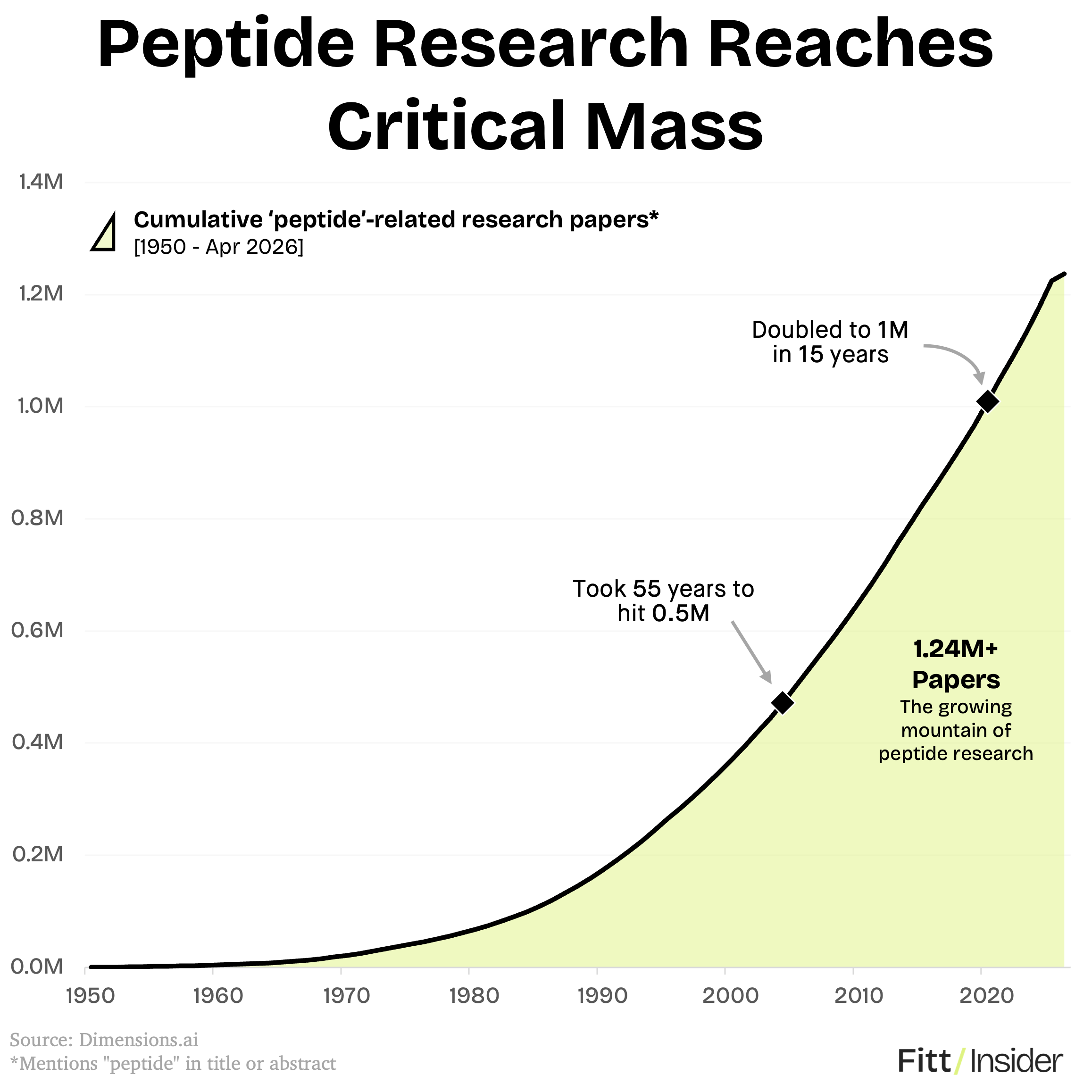
Scaling up. For some of the 12 drugs in question, the hype is catching up to the science. BPC-157 dates to the ’90s, GHK-Cu to the ’70s, and Thymosin alpha-1 is approved in 35+ countries. Behind the scenes, peptide research now exceeds 1.2M papers — doubling in the last 15 years.
Broken incentives. Economics, not evidence, keep peptides in limbo. Despite forming an $80B market, many compounds remain unapproved. Naturally occurring and hard to patent, companies see $100M+ trials as a nonstarter.
Proof point. From GLP-1 crackdowns to compounded alternatives, regulators and operators are already clashing over access. RFK Jr. is pushing expansion, while critics warn policy is moving ahead of clinical proof and safety.
But for wellness consumers, restrictions didn’t stop demand — they rerouted it to medspas, telehealth platforms, and gray-market suppliers.
On cue. Regulated compounding could redirect usage back into the system, and vertical integration could create power players — with Hims and Noom acquiring 503A facilities and Protocole building trust with physician-guided stacking.
Punchline: With decades of research and a multi-billion-dollar gray market in motion, the peptide revolution is already here; the FDA is just catching up.