Psychedelics are moving from fringe to frontline.
What’s happening: A new executive order aims to accelerate research into psychedelics for mental health.
All clear. Targeting compounds like psilocybin, MDMA, and ibogaine, the order reduces regulatory barriers, fast-tracks clinical pathways, and allocates $50M in public funding for state-level studies. It also introduces priority FDA review and expanded early-access pathways.
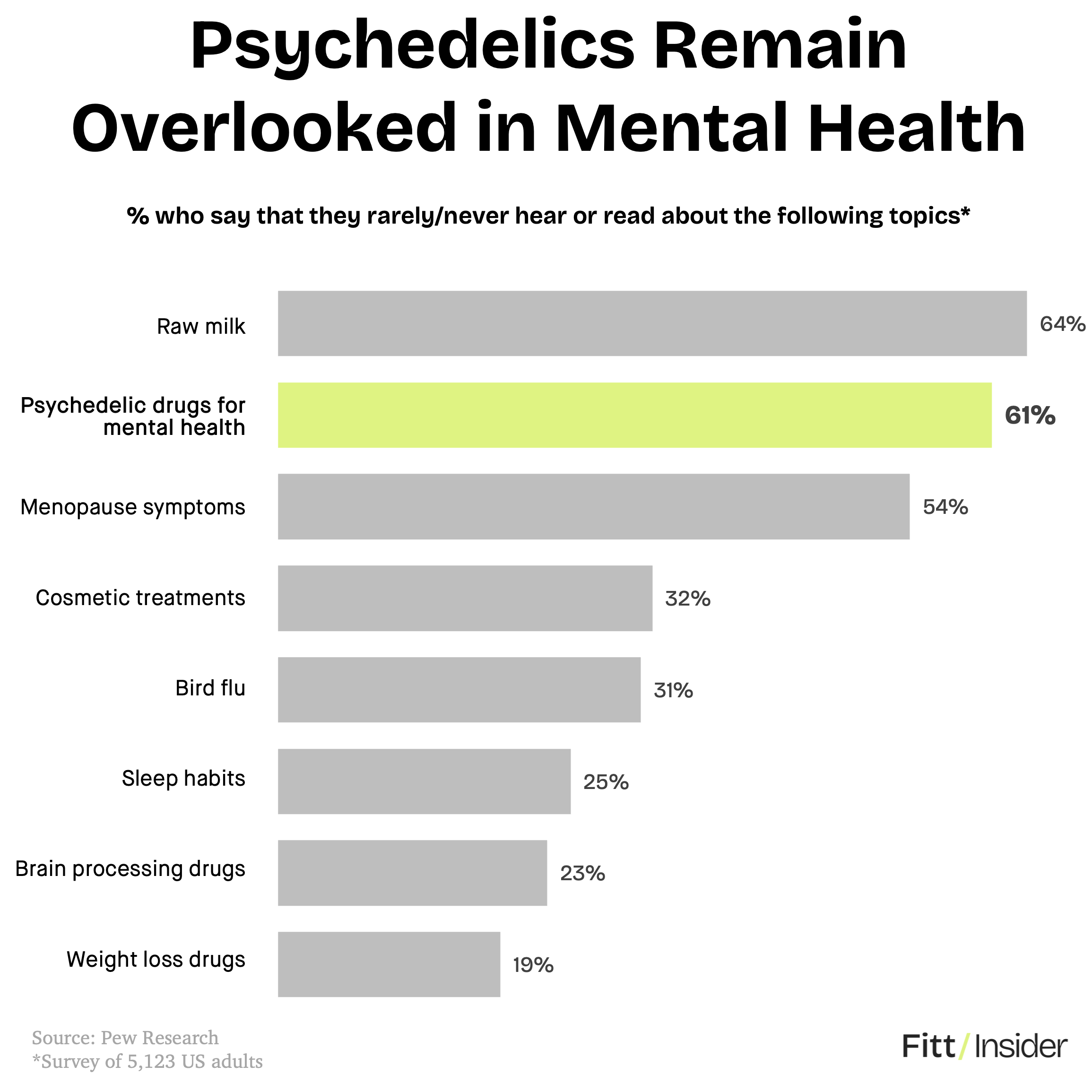
Breaking through. Restricted and stigmatized as Schedule I drugs, these substances are gaining traction as treatments for depression, PTSD, and addiction.
Validating the hype, trials show MDMA-assisted therapy driving 86% improvement in PTSD, with 71% of patients reaching remission. Psilocybin is also showing progress in combating treatment-resistant depression. A head start, Texas approved $50M last year to research ibogaine for SUD and TBIs.
Momentum. As capital, policy support, and cultural acceptance align, psychedelics are drawing in investors and operators. Drug developers like Compass Pathways and AtaiBeckley are advancing clinical pipelines, while platforms like Mindbloom’s DTC ketamine therapy expand access.
Takeaway: Psychedelics are being reclassified. As clinical and cultural acceptance grow, they could become a new pillar of mental healthcare.






